Under development of antibody-based therapeutics suitable to indication and target


-
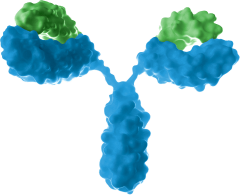
Monoclonal antibody
- Targeting novel tumor associated antigens
-
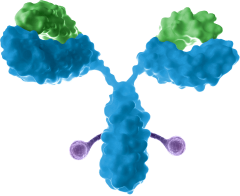
Antibody-drug conjugates, ADCs
- Improved efficacy and safety
- Toxins, Radioisotope, etc.
-
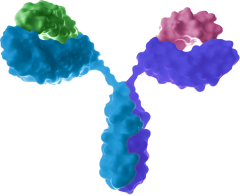
Bispecific antibody
- Synergistical effects by dual targeting
- Overcome drug resistance and improve efficacy
-
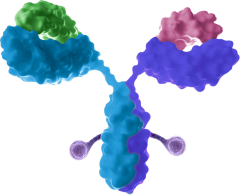
Bispecific antibody-drug conjugate
- Improved cancer cell selectivity, internalization, payload release
The potential of ADCs to overcome the limitation of current cancer therapies
-
Next-generation drug maximizing the advantages of 1st to 3rd generation of anti-cancer therapies
ADC enables to overcome the unwanted non-specific killing of normal and cancer cells caused by traditional chemotherapy
-
Providing new treatment opportunities for cancer patients with limited options
Safe and maximized anti-cancer effects through selective delivery of drugs to target cancer cells
Strengths in the development of ADCs by antibody asset companies
-
ADC approvals and clinical studies
-
14 approved drugs and over 300 clinical trials ongoing in 2023
Experience has been accumulated in linker and payload development.
-
-
Securing antibodies for disease-selective targets: The key to successful ADC development
-
Patents for approved ADC linkers and payloads are expiring
-
Opportunities for ADC development by securing novel antibodies
ED2 pipeline is based on our proprietary tumor targeting antibody combined with proven linker and payloads
-
-
Strengths in the development of ADCs by antibody asset companies
-
First in Class drug development
Use linkers and payloads that have already been confirmed to be safe for new antibodies -
Low biological risk, low competition, and low probability of clinical trial failure
-
